 Your new post is loading...
 Your new post is loading...
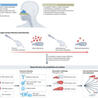
Researchers at Karolinska Institutet have shown that nasal drops with IgA antibodies can protect mice from SARS-CoV-2 infection. The results imply a new way to protect individuals at high risk from different variants of the SARS-CoV-2 virus and possibly other infections. The study is published in PNAS.
In this issue of The Journal of Rheumatology, Lee et al present an interesting editorial claiming that seronegative Sjögren syndrome (SS) is a forgotten entity.1 They provide convincing arguments supporting this assertion.
Numerous pathogens can infect the olfactory tract, yet the pandemic caused by SARS-CoV-2 has strongly emphasized the importance of the olfactory mucosa as an immune barrier. Situated in the nasal passages, the olfactory mucosa is directly exposed to the environment to sense airborne odorants; however, this also means it can serve as a direct route of entry from the outside world into the brain. As a result, olfactotropic infections can have serious consequences, including dysfunction of the olfactory system, CNS invasion, dissemination to the lower respiratory tract, and transmission between individuals. Recent research has shown that a distinctive immune response is needed to protect this neuronal and mucosal tissue. A better understanding of innate, adaptive, and structural immune barriers in the olfactory mucosa is needed to develop effective therapeutics and vaccines against olfactotropic microbes such as SARS-CoV-2. Here, we summarize the ramifications of SARS-CoV-2 infection of the olfactory mucosa, review the subsequent immune response, and discuss important areas of future research for olfactory immunity to infectious disease.
The gut microbiome has important roles in host metabolism and immunity, and microbial dysbiosis affects human physiology and health. Maternal immunity and microbial metabolites during pregnancy, microbial transfer during birth, and transfer of immune factors, microorganisms and metabolites via breastfeeding provide critical sources of early-life microbial and immune training, with important consequences for human health. Only a few studies have directly examined the interactions between the gut microbiome and the immune system during pregnancy, and the subsequent effect on offspring development. In this Review, we aim to describe how the maternal microbiome shapes overall pregnancy-associated maternal, fetal and early neonatal immune systems, focusing on the existing evidence and highlighting current gaps to promote further research. This Review discusses how the maternal gut microbiome in pregnancy influences the developing immune system and proposes avenues for future research.
In this Review, Danne and colleagues describe the roles of neutrophils in inflammatory bowel disease, as well as their functions in host–microbiota interactions.
Review in Nature Reviews Gastroenterology and Hepatology explores how the maternal microbiome during pregnancy influences the immune systems of the mother, fetus, and neonate, highlighting its impact on long-term health outcomes and the development of pregnancy complications.
Commensal bacteria, such as Bacteroides fragilis, triggers type I IFN and IL-27 in DCs to influence Tregs through IL27RA signaling. The IFN gene signature in Tr
Compared to intramuscular vaccines, nasally administered vaccines have the advantage of inducing local mucosal immune responses that may block infection and interrupt transmission of respiratory pathogens. Live attenuated influenza vaccine (LAIV) is effective in preventing influenza in children, but a correlate of protection for LAIV remains unclear. Studying young adult volunteers, we observe that LAIV induces distinct, compartmentalized, antibody responses in the mucosa and blood. Seeking immunologic correlates of these distinct antibody responses we find associations with mucosal IL-33 release in the first 8 hours post-inoculation and divergent CD8+ and circulating T follicular helper (cTfh) T cell responses 7 days post-inoculation. Mucosal antibodies are induced separately from blood antibodies, are associated with distinct immune responses early post-inoculation, and may provide a correlate of protection for mucosal vaccination. This study was registered as NCT04110366 and reports primary (mucosal antibody) and secondary (blood antibody, and nasal viral load and cytokine) endpoint data. Nasally delivered live attenuated influenza vaccines (LAIV) have been shown to be effective in vaccine trials yet immune responses are mostly measured in blood. Here the authors report a clinical trial in young adults and measure immune responses in the mucosa and blood to identify compartmentalised responses.
|
Social media platforms are used for support and as resources by people from the endometriosis community who are seeking advice about diagnosis, education, and disease management. However, little is known about the scientific accuracy of information circulated on Instagram about the disease.
Our bodies are in continuous contact with diverse microorganisms, with most exposure occurring at epithelial sites. The skin and digestive tract epithelia are t
The complex and highly dynamic lung immune compartment.
Early human lung immune cell development and its role in epithelial cell fate | Science Immunology…
Scientists proposed a new nano-micro composite delivery concept for vaccines- combines biodegradable microspheres with protein nanoparticles.
Pneumonia is a common acute respiratory infection that affects the alveoli and distal airways; it is a major health problem and associated with high morbidity and short-term and long-term mortality in all age groups worldwide. Pneumonia is broadly divided into community-acquired pneumonia or hospital-acquired pneumonia. A large variety of microorganisms can cause pneumonia, including bacteria, respiratory viruses and fungi, and there are great geographical variations in their prevalence. Pneumonia occurs more commonly in susceptible individuals, including children of <5 years of age and older adults with prior chronic conditions. Development of the disease largely depends on the host immune response, with pathogen characteristics having a less prominent role. Individuals with pneumonia often present with respiratory and systemic symptoms, and diagnosis is based on both clinical presentation and radiological findings. It is crucial to identify the causative pathogens, as delayed and inadequate antimicrobial therapy can lead to poor outcomes. New antibiotic and non-antibiotic therapies, in addition to rapid and accurate diagnostic tests that can detect pathogens and antibiotic resistance will improve the management of pneumonia. Pneumonia is a respiratory infection of the distal airways; it can be acquired in the community or in the hospital, and it can be caused by several types of bacteria, viruses, fungi and other pathogens.
🔬 Launch of the MUCOVID study 🔬
What is the MUCOVID study?
Lovaltech's mission is to study the immune response of our mucosa, the gateway to most…
Although fermentation probably originally developed as a means of preserving food substrates, many fermented foods (FFs), and components therein, are thought to have a beneficial effect on various aspects of human health, and gastrointestinal health in particular. It is important that any such perceived benefits are underpinned by rigorous scientific research to understand the associated mechanisms of action. Here, we review in vitro, ex vivo and in vivo studies that have provided insights into the ways in which the specific food components, including FF microorganisms and a variety of bioactives, can contribute to health-promoting activities. More specifically, we draw on representative examples of FFs to discuss the mechanisms through which functional components are produced or enriched during fermentation (such as bioactive peptides and exopolysaccharides), potentially toxic or harmful compounds (such as phytic acid, mycotoxins and lactose) are removed from the food substrate, and how the introduction of fermentation-associated live or dead microorganisms, or components thereof, to the gut can convey health benefits. These studies, combined with a deeper understanding of the microbial composition of a wider variety of modern and traditional FFs, can facilitate the future optimization of FFs, and associated microorganisms, to retain and maximize beneficial effects in the gut. Fermented foods have gained renewed popularity owing to their health-promoting potential. This Review discusses the mechanisms underlying the benefits of fermented foods in gut health and disease, highlighting how specific fermented food microorganisms, food components and bioactive compounds exert their effects. A future outlook on research is also detailed.
Jean Barbeau, microbiologiste, Responsable de la prévention et du Contrôle des infections, FMD, Université de Montréal. Nous connaissons très bien les vaccins donnés par une injection dans le muscle de l'épaule. Ce type de vaccins nous a permis, dès la fin 2020, d'assurer une excellente protection contre SRAS-CoV-2. Pour comprendre un peu mieux l'immunisation (vaccination)…
|
 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...




















at last, mucosal immunity on stage